 The delivery of paracetamol from a glass vial allows air to enter the intravenous giving set risking venous air embolus. Paracetamol is already presented in bags which don’t entrain air and is used in this form without issue in many hospitals.
The delivery of paracetamol from a glass vial allows air to enter the intravenous giving set risking venous air embolus. Paracetamol is already presented in bags which don’t entrain air and is used in this form without issue in many hospitals.
The distraction afforded through keeping a watchful eye on glass vials of paracetamol to avoid air entrainment may detract from patient care. When air does enter the giving set it requires time and an additional syringe to remove it.
Perhaps it would be ideal if worldwide intravenous paracetamol were only provided in bags.
Help:
– If you know of any institution supplying intravenous paracetamol in glass vials provide them with a copy of this post.
– We should create a list of manufacturers of intravenous paracetamol in glass vials (e.g. Fresenius Kabi) and write a letter to their CEO requesting they consider changing to supply it in bags. Anyone who sees a manufacturer supplying intravenous paracetamol in glass vials should add to this list.
– We should develop a list of suppliers of intravenous paracetamol in bags for distribution to those institutions currently supplying it in vials.
This issue may seem trivial however it provides a great example of the culture we have been nurtured in.
Perhaps we put up with these issues because we can and doing so satisfies our egos. But should we? Shouldn’t we be doing everything we can to remove unnecessary hazards. Perhaps current healthcare culture doesn’t support this attitude.
Perhaps it would be a good exercise for hospital managers to truly understand why front line staff haven’t brought these issues up with them earlier in a sufficient manner to have them resolved.
That this unnecessary hazard exists in our hospitals is testimony that we’ve become too numbed to improve our front line environments. Front line staff have been nurtured in healthcare with a paucity of frameworks to improve working conditions.
That there are no systems for reporting hazards conveys a message that front line staff should just put up with their work environments.
As healthcare stands few will be criticised if we maintain the status quo, keep quiet about the hazards yet present the adverse events which occur because of their presence into inneffective opaque reporting systems. As a result the same adverse events repeat again and again and again.
If we are to improve we need the frameworks to allow it. Matthew Syed in his book, Black Box Thinking, points out that we need the ability to improve what might seem like the smallest of issues – the ‘one percenters’. He goes on to demonstrate this is how the British Skye racing team has used this ability to come from nowhere to dominate the Tour de France.
Despite health care relying on ‘root cause analysis’ to review major adverse events there is never one root cause. Adverse events occur because of a series of ‘holes in a piece of cheese’ – we need to be awakened from our slumber in healthcare and start to remove all of the unnecessary holes however trivial they might seem.
Welcome to the PatientSafe Network.
Subnote:
An important issue which has been raised by several readers is that of bags of paracetamol being mistaken for other 100ml fluid bags e.g. saline, metronidazole. This is a significant issue which we must also respect and do everything we can to overcome. Below are images of paracetamol fluid bags which come pre-packaged in more discernible outer bags – still not perfect. If others have images of better presentations of paracetamol please forward them on:
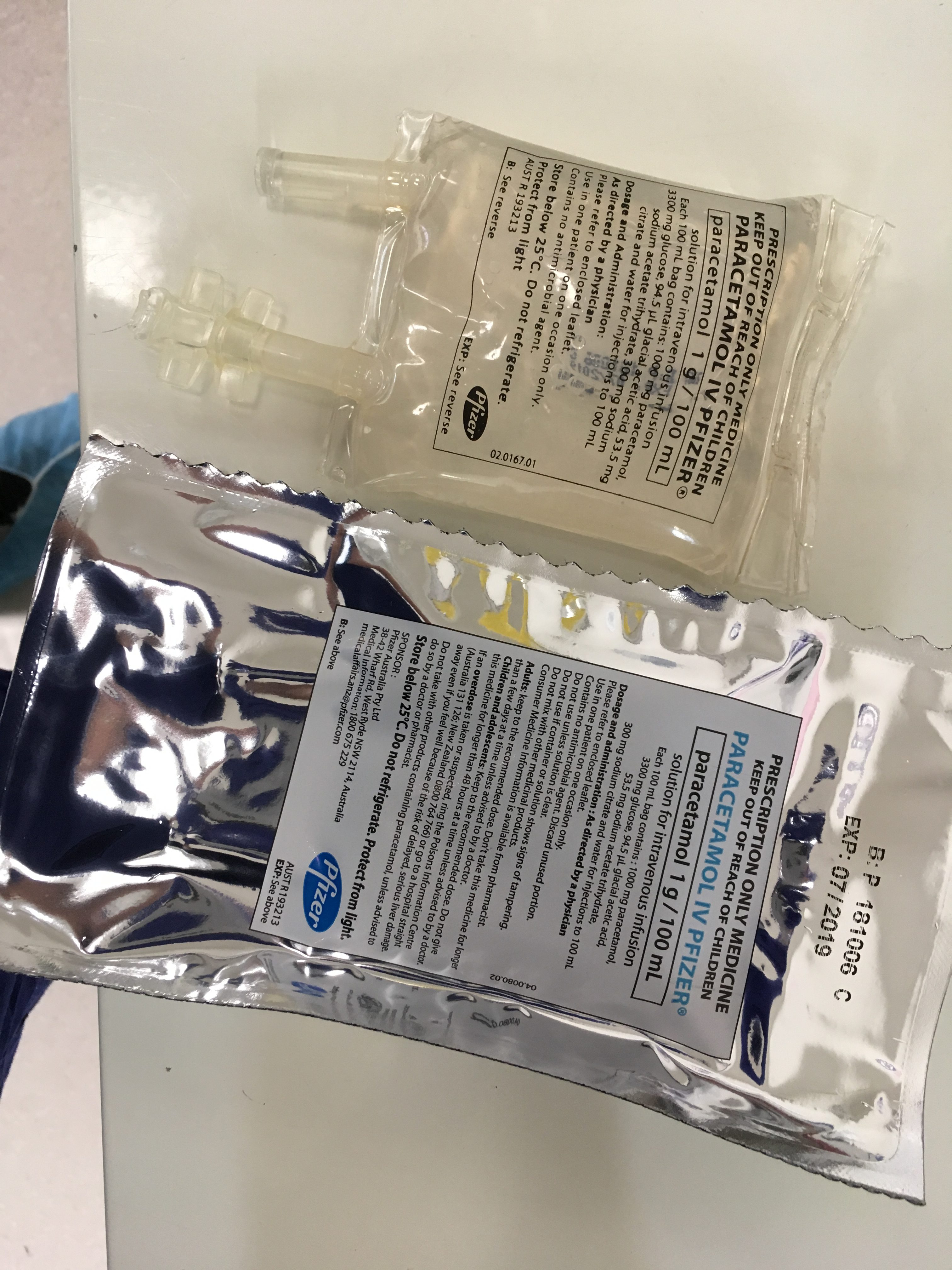
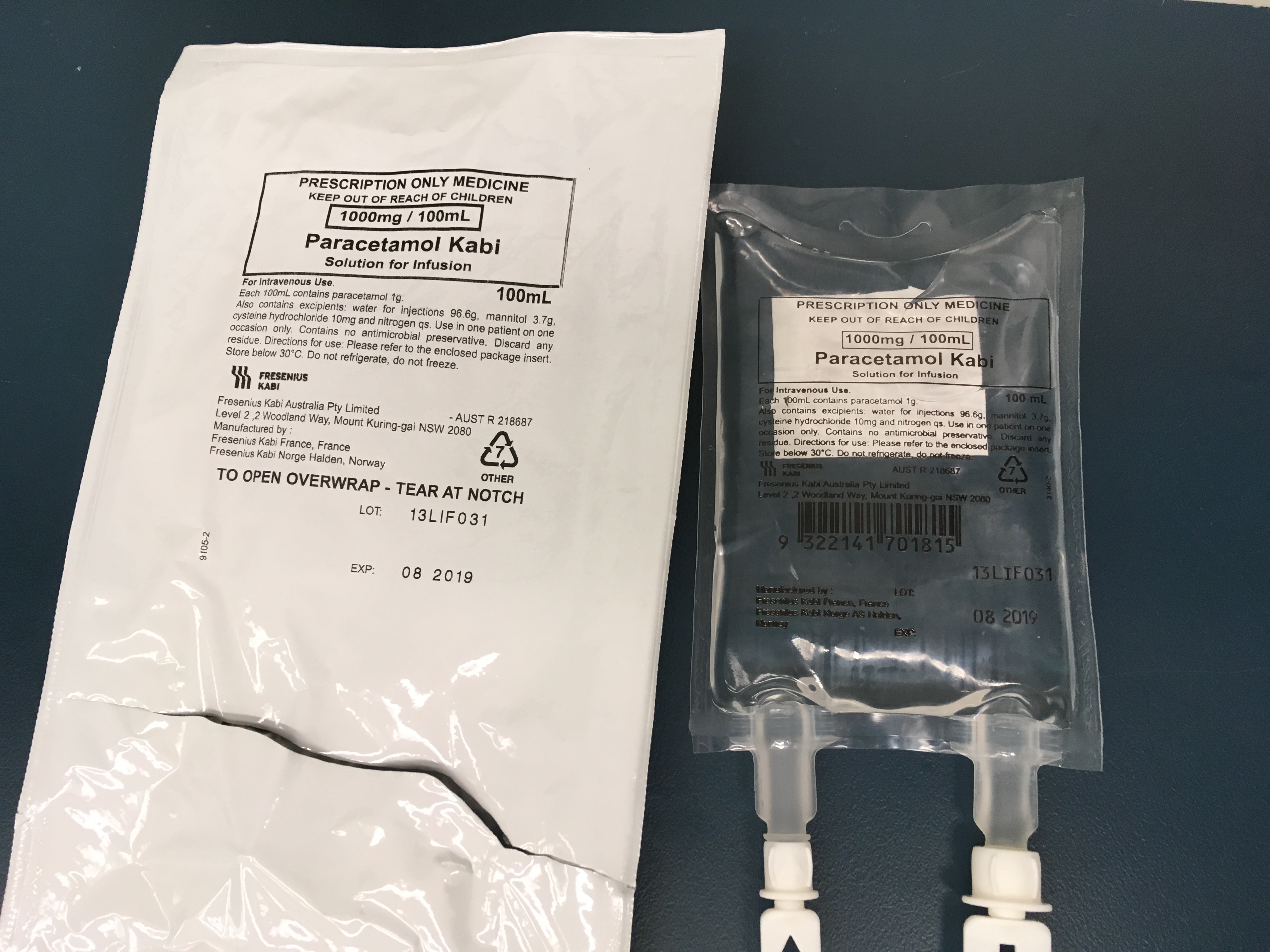
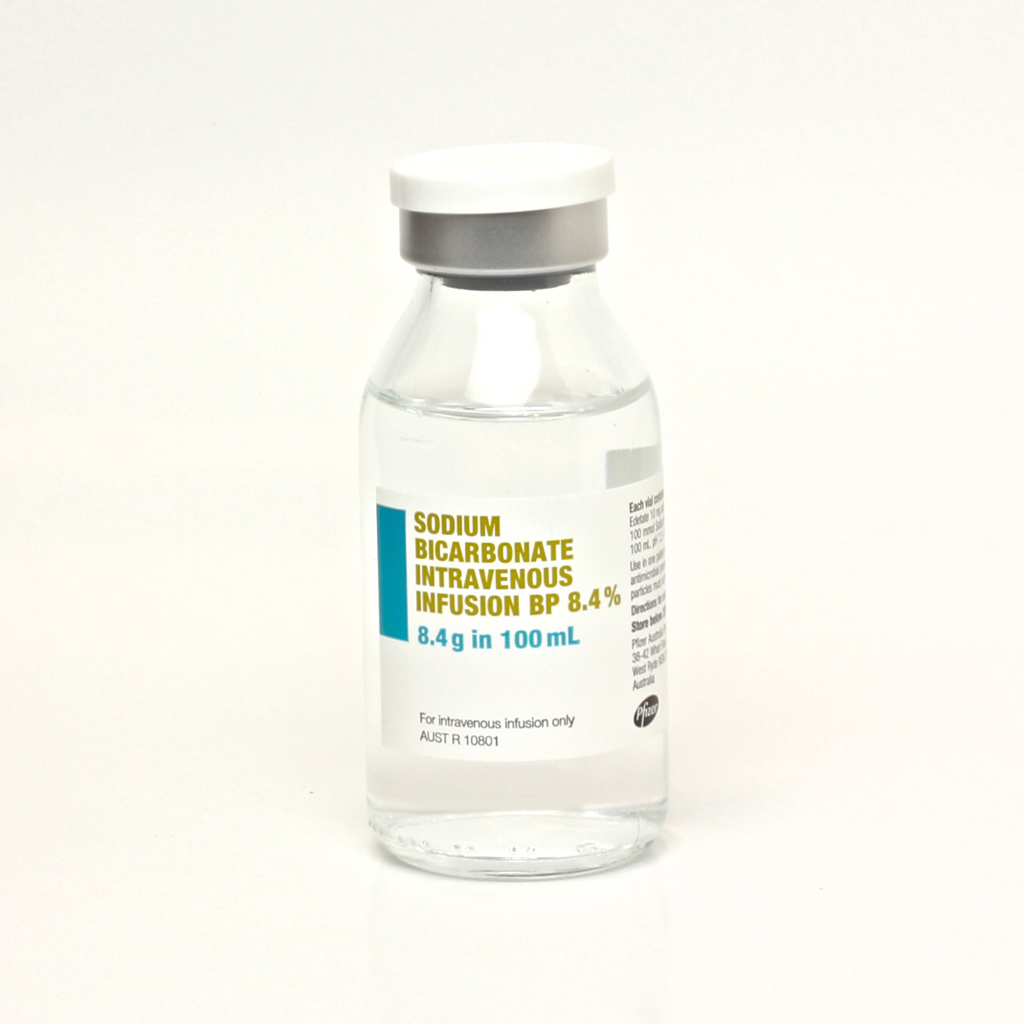
Further paracetamol vials are not immune to being mistaken for other vials. There have been several reports of sodium bicarbonate vials being mistaken for paracetamol vials:

11 Comments